New principles published to improve public confidence in access and use of data for health research through Trusted Research Environments
8 December 2021
UK Health Data Alliance Paper – “Principles and Best Practices for Trusted Research Environments” including supporting foreword from NHSX – sets out approach for a trustworthy ecosystem of data access for health research
The UK Health Data Research Alliance has today published a paper for organisations that have and are building Trusted Research Environments (TREs) for health data.
The level of interest in Trusted Research Environments and their benefits has grown substantially in the last 12 months, due to their potential to transform the ability to carry out research across the health system in a way that has the confidence and trust of patients, public, clinicians and researchers.
Public awareness of the use of health data for research has also increased substantially as a result of the critical role of research and health data in response to Covid-19 pandemic, raising legitimate questions around transparency and security in their use.
The paper acts as a guide for UK data custodians and other organisations involved in data sharing and information governance both within and beyond the health sectors. It identifies and provides principles, approaches and guidelines for data sharing and linkage practice within TREs, as well as examples of best practices drawn from the experiences of colleagues working in the Alliance and internationally.
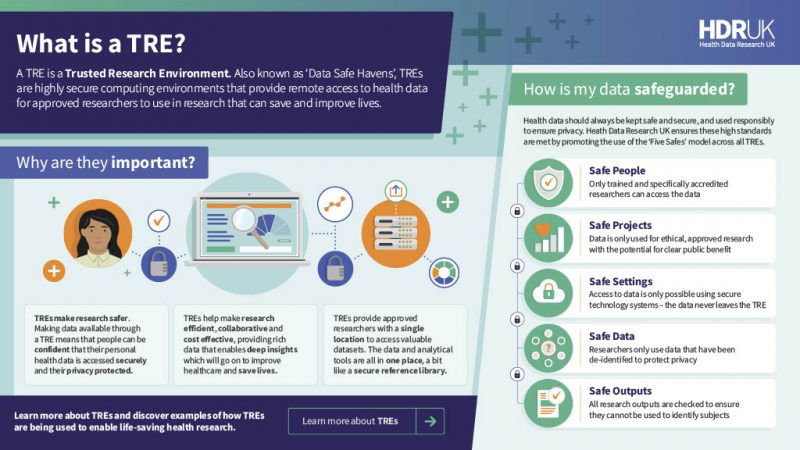
The guidelines and principles in the paper are structured around the “Five Safes” framework for the access of health data – safe people, safe projects, safe settings, safe data, safe outputs. The paper also sets out principles for the federation of Trusted Research Environments – how they can be linked, to substantially enhance the scale of secure and trustworthy data linkage and research.
The paper is published by the UK Health Data Research Alliance (“The Alliance”) in its role as an independent partnership of leading healthcare and research organisations united to establish best practice for the ethical use of UK health data for research at scale. It leverages HDR UK’s in-house expertise in data engineering, as well as input from Alliance members, other partners, patients and the HDR UK Public Advisory Board, building on the Green Paper published in July 2020.
David Seymour, Alliance Executive Director, Health Data Research UK:
“Trusted Research Environments are an important tool in how we advance scientific discovery through ethically robust research. This paper represents our commitment to their successful development, so that, as a sector, we can align on standards that protect privacy and improve transparency in the use of patient data for research.”
Simon Madden, Director of Data Policy & Covid Pass Policy, NHSX
“The UK has some of the richest health data in the world. Combined with our research expertise in the NHS, academia and life sciences industries, we have the opportunity to use this data at scale for patient benefit. This paper will help us ensure this is done in a secure, trustworthy way.”
Read the full paper: https://doi.org/10.5281/zenodo.5767586
What is a Trusted Research Environment (TRE)?
Also known as “Data Safe Havens” or “Secure Data Environments”, TREs are highly secure computing environments that provide remote access to health data for approved researchers to use in research, which will go on to improve healthcare and save lives.
TREs provide researchers with a single location to access valuable datasets. Data and analytical tools are located in one place, similar to a secure reference library. Rather than extracts of individual level data being ’released’, TREs provide access to a secure analytics environment (“safe setting”) so researchers bring analysis (such as algorithms) to the data. Making data available through a TRE provides confidence to patients and the public that their personal health data is accessed securely, and their privacy is protected.
TREs also help make research efficient, collaborative and cost effective – it avoids the costs of transferring and storing duplicates of increasingly large datasets, particularly imaging and genomic data. It also avoids the liabilities for researchers from having to ensure security of downloaded datasets.
Next steps
HDR UK and NHSX are progressing joint workstreams to develop and codify best practice for TREs and TRE for NHS data (discussed in the foreword of the paper). There is ongoing work to define requirements for implementing and operating TREs, such as accreditation, as well as the need to pilot technical implementations of federation.
The Data and Analytics Research Environments UK (DARE UK) programme of UKRI, ADR UK and HDR UK was launched in July 2021 to design and deliver a co-ordinated, national data research infrastructure that demonstrates trustworthiness and supports research at scale for wider public good (as well as healthcare research).
Media enquiries:
Joel Beckman, Head of Communications, Health Data Research UK
Anna Steere, Deputy Head of Communications NHS X / NHS England
Data Officers/Custodians:
If you are a Data Officer/Custodian and would like to find out how the UK Health Data Alliance can support your work, please contact Paola Quattroni.
Further information:
Health Data Research UK – Trusted Research Environments
The UK Health Data Research Alliance – Aligning approach to Trusted Research Environments
HDR UK Learning – “Working in a TRE”
NHS X – Joining The Dots blog by Simon Madden and Catherine Pollard:
Understanding Patient Data – “The What and Why of trusted research environments” blog by Professor Cathie Sudlow
Examples of Trusted Research Environments in the UK
There are many TREs operating successfully, both for healthcare data and other potentially sensitive data. The responses to the Covid-19 pandemic have accelerated this shift, with a number of other organisations investigating setting up TREs, such as Our Future Health.
A sample of existing TREs for health research:
- Scotland Data Safe Haven programme
- UK Secure eResearch Platform in Wales
- Genomics England Research Environment
- Office of National Statistics Secure Research Service
- UK Data Service Secure Lab
- NHS Digital TRE for England
- OpenSAFELY
Trusted Research Environments “Principles and Best Practice”
Key milestones in co-creation
| June 2019 | Workshop at HDR UK “One Institute” Meeting |
| September 2019 | Dedicated workstream established to produce Green Paper |
| January 2020 | Draft Green Paper presented to UK Health Data Alliance Board |
| February 2020 | Working session at HDR Alliance Symposium |
| April 2020 | Presentation of draft Green Paper to HDR Public Advisory Board |
| May 2020 | Public consultation on Green Paper launched |
| July 2020 | Updated Green Paper published with next steps |
| December 2020 | Working session at UK HDR Alliance Symposium (85 attendees) |
| July 2021 | Drafting of paper commences |
| December 2021 | “Principles and Best Practice” paper published, including supporting foreword from NHSX |
Recent policy context (with relevance for Trusted Research Environments)
- Data saves lives: Reshaping health and social care with data – draft Data Strategy for Health and Social Care – launched by DHSC and NHSX (June 2021)
- Department of Health review into use of health data for research and analysis (“The Goldacre Review”)
- GP Data for Planning and Research – update from Secretary of State, including commitment on TRE)
- Saving and improving lives: the future of UK clinical research delivery
- Genome UK 2021-22 implementation plan
- Life Sciences Vision
The UK Health Data Research Alliance
The UK Health Data Research Alliance is an independent partnership of leading healthcare and research organisations united to establish best practice for the ethical use of UK health data for research at scale.
The Alliance develops and co-ordinates the adoption of tools, techniques, conventions, technologies, and designs that enable the use of health data in a trustworthy and ethical way for research and innovation. By combining expertise and a shared commitment to work collaboratively, the Alliance is helping researchers to answer some of the most difficult questions and address the most important health challenges faced in the UK
The Alliance is co-ordinated by Health Data Research UK and part of the UK-wide capability for the responsible use of health data for research and innovation, funded by the Industrial Strategy Challenge Fund (ISCF).
Health Data Research UK
Health Data Research UK (HDR UK) is the national institute for health data science. Our mission is to unite the UK’s health data to enable discoveries that improve people’s lives.
HDR UK was established in 2018 to support research on health data at scale to advance our understanding of disease and enable new discoveries that will ultimately improve health and care.
We have established national research programmes that use data at scale, and we are building an infrastructure to enable the responsible access and analysis of this data.